Proven for 15 Years
> Proven Simplicity
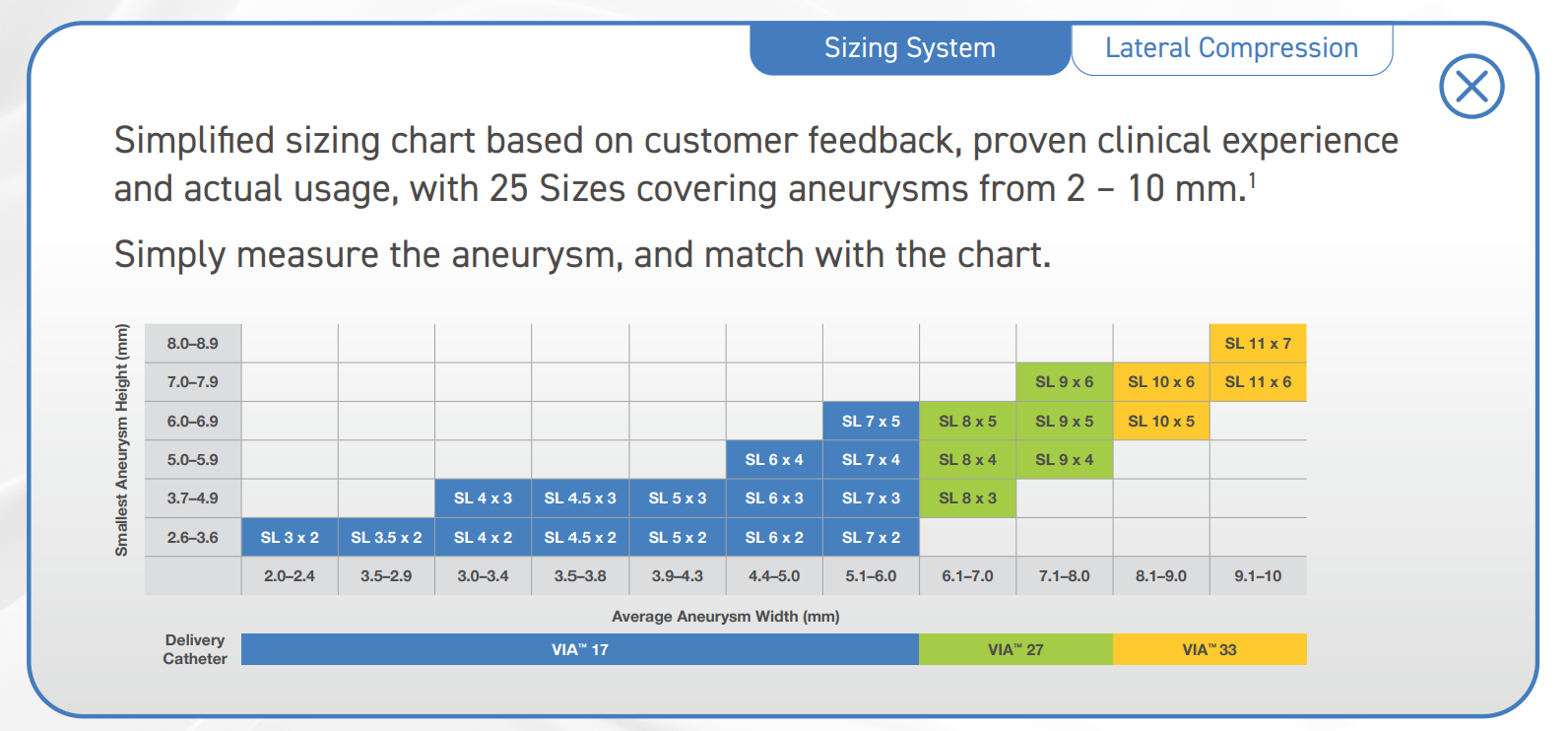
Sizing system
2.7%
Mean % of cases with adjunctive device implanted4,5,6
Mean rate of successful WEB device placement4,5,7
Reduces procedure time and radiation exposure
20.1min
Mean WEB device procedure time4
48%
Reduction in radiation time8
> Proven Safety and Durability
Proven Safety
0%
Bleeding or rebleeding
in GCP studies2,3,5,6,7,9
0%
WEB device related mortality
in GCP studies2,3,5,6,7,9
0%
WEB™ related morbidity
in GCP studies2,3,5,6,7,9
Proven Short-Term Efficacy
84.6%
Mean adequate occlusion at angiographic
follow up at 1 year in GCP studies9,10,11
91.1%
Mean adequate oclusion at 3-12 months follow up
in independent European Center experiences12,13,14
5.2%
Mean retreatment rate
at 1 year9,10,11,15
Proven Long-Term Durability
83.6%
Mean adequate occlusion at
5 years angiographic follow up2,3
84.6% to 87.2%
Adequate occlusion evolution in
1 to 5 years follow-up in WEB-IT3
87.8%
Occlusion remained stable or improved in
1 to 5 years follow up in WEBCAST1 & 22
> Proven for Ruptured
2
coverage areas1
1.7%
aneurysm meta-analysis
at mid-term follow up8
86.7%
ruptured aneurysm GCP studies10,11
NO DAPT
aneurysms treated with
WEB device12
9.5% vs 23.2%
complications vs stent assisted coiling (SAC)13
94%
a mean follow up of
51 months14

PROVEN SIMPLICITY
References
1. Data on File, Terumo Neuro
4. Fiorella D, Molyneux A, Coon A, et al. Demographic, procedural and 30-day safety results from the WEB Intra-saccular Therapy Study (WEB-IT). J Neurointerv Surg 2017.
5. Spelle L, Costalat V, Caroff J et al. Clinical Evaluation of WEB 17 device in intracranial aneurysms (CLEVER): procedural, 30-day and 1-year safety results for ruptured and unruptured aneurysms. J NeuroIntervent Surg 2023.
6. Spelle L, Herbreteau D, Caroff J et al. CLinical Assessment of WEB device in Ruptured aneurYSms (CLARYS): results of 1-month and 1-year assessment of rebleeding protection and clinical safety in a multicenter study. J Neurointerv Surg 2022.
7. Pierot L, Moret J, Barreau X, et al. Safety and efficacy of aneurysm treatment with WEB in the cumulative population of three prospective, multicenter series. J Neurointerv Surg 2018.
8. Rai A, Turner R , Brotman R et al. Comparison of operating room variables, radiation exposure and implant costs for WEBTM versus stent assisted coiling for treatment of wide neck bifurcation aneurysms. Interv Neuroradiol. 2021.
9. DelgadAlmandoz J, Kayan Y, Copelan A et al. Lateral Compression is the Strongest Independent Predictor of Aneurysm Occlusion After Endovascular Treatment of Intracranial Aneurysms With the Woven EndoBridge Device. Neurosurgery Practice 2023.
10. Mihalea C, Caroff J, Pagiola I et al. Safety and efficiency of the fifth generation Woven EndoBridge device: technical note. Neurinterventional Surgery 2019.
11. Van Rooij S.B.T., Peluso J.P., Sluzewski M. et al.The New Low-Profile WEB 17 System for Treatment of Intracranial Aneurysms: First Clinical Experiences. American Journal of Neuroradiology 2018.
12. Goertz L , Liebig T, Siebert E et al Low-Profile Intra-Aneurysmal Flow Disruptor WEB 17 versus WEB Predecessor Systems for Treatment of Small Intracranial Aneurysms: Comparative Analysis of Procedural Safety and Feasibility. American Journal of Neuroradiology 2019.
PROVEN SAFETY & DURABILITY
References
1. Data on File, Terumo Neuro.
2. Pierot L, Szikora I, Barreau X, et al. Aneurysm treatment with the Woven EndoBridge (WEB) device in the combined population of two prospective, multicenter series: 5-year follow up. J Neurointerv Surg. 2023.
3. Fiorella D, Molyneux A, Coon A, et al. Safety and effectiveness of the Woven EndoBridge (WEB) system for the treatment of wide necked bifurcation aneurysms: final 5 year results of the pivotal WEB Intra-saccular Therapy study (WEB-IT). J Neurointerv Surg 2023.
5. Spelle L, Costalat V, Caroff J et al. Clinical Evaluation of WEB 17 device in intracranial aneurysms (CLEVER): procedural, 30-day and 1-year safety results for ruptured and unruptured aneurysms. J NeuroIntervent Surg 2023.
6. Spelle L, Herbreteau D, Caroff J et al. CLinical Assessment of WEB device in Ruptured aneurYSms (CLARYS): results of 1-month and 1-year assessment of rebleeding protection and clinical safety in a multicenter study. J Neurointerv Surg 2022.
7. Pierot L, Moret J, Barreau X, et al. Safety and efficacy of aneurysm treatment with WEB in the cumulative population of three prospective, multicenter series. J Neurointerv Surg 2018.
9. Arthur AS, Molyneux A, Coon A, et al. The safety and effectiveness of the Woven EndoBridge (WEB) system for the treatment of wide-necked bifurcation aneurysms: final 12 month results of the pivotal WEB Intrasaccular Therapy (WEB-IT) Study. J Neurointerv Surg 2019.
10.Spelle L, Costalat V, Caroff J et al. CLinical EValuation of WEBTM 17 device in intracranial aneuRysms (CLEVER): 1 year effectiveness results for ruptured and unruptured aneurysms. J NeuroIntervent Surg 2024.
11. Spelle L, Herbreteau D, Caroff J et al. CLinical Assessment of WEBTM Device in Ruptured AneurYSms (CLARYS): 12 month angiographic results of a multicenter study. J NeuroIntervent Surg. 2023.
12. Kovan Rooij SBT, Peluso JP, Sluzewski M et al. The New Low-Profile WEB 17 System for Treatment of Intracranial Aneurysms: First Clinical Experiences. American Journal of Neuroradiology 2018.
13. Maurer C, Konig I, Berlis A et al. Two-Center Experience in the Endovascular Treatment of Intracranial Aneurysms Using the Woven EndoBridge 17 Device Including Midterm follow up Results: A Retrospective Analysis. American Journal of Neuroradiology 2019.
14. Konig I, Maurer C, Berlis A et al. Treatment of Ruptured and Unruptured Intracranial Aneurysms with WEB 17 Versus WEB 21 Systems : Comparison of Indications and Early Angiographic Outcomes. Clin Neuroradiol 2021.
15. M A Essibayi, G Lanzino , W Brinjikji et al. Safety and Efficacy of the Woven EndoBridge Device for Treatment of Ruptured Intracranial Aneurysms: A Systematic Review and Metanalysis. American Journal of Neuroradiology 2021.
PROVEN FOR RUPTURED
References
1. Data on File, Terumo Neuro.
2. Pierot L, Szikora I, Barreau X, et al. Aneurysm treatment with the Woven EndoBridge (WEB) device in the combined population of two prospective, multicenter series: 5-year follow up. J Neurointerv Surg. 2022.
3. Fiorella D, Molyneux A, Coon A, et al. Safety and effectiveness of the Woven EndoBridge (WEB) system for the treatment of wide necked bifurcation aneurysms: final 5 year results of the pivotal WEB Intra-saccular Therapy study (WEB-IT). J Neurointerv Surg 2023.
4. Fiorella D, Molyneux A, Coon A, et al. Demographic, procedural and 30-day safety results from the WEB Intra-saccular Therapy Study (WEB-IT). J Neurointerv Surg 2017.
5. Spelle L, Costalat V, Caroff J et al. Clinical Evaluation of WEB 17 device in intracranial aneurysms (CLEVER): procedural, 30-day and 1-year safety results for ruptured and unruptured aneurysms. J NeuroIntervent Surg 2023.
6. Spelle L, Herbreteau D, Caroff J et al. CLinical Assessment of WEB device in Ruptured aneurYSms (CLARYS): results of 1-month and 1-year assessment of rebleeding protection and clinical safety in a multicenter study. J Neurointerv Surg 2022.
7. Pierot L, Moret J, Barreau X, et al. Safety and efficacy of aneurysm treatment with WEB in the cumulative population of three prospective, multicenter series. J Neurointerv Surg 2018.
8. Rai A, Turner R , Brotman R et al. Comparison of operating room variables, radiation exposure and implant costs for WEBTM versus stent assisted coiling for treatment of wide neck bifurcation aneurysms. Interv Neuroradiol. 2021.
9. Essibayi MA, Lanzino G, Brinjikji W et al. Safety and Efficacy of the Woven EndoBridge Device for Treatment of Ruptured Intracranial Aneurysms: A Systematic Review and Metanalysis. American Journal of Neuroradiology 2021.
10. Spelle L, Costalat V, Caroff J et al. CLinical EValuation of WEBTM 17 device in intracranial aneuRysms (CLEVER): 1-year effectiveness results for ruptured and unruptured aneurysms. J NeuroIntervent Surg 2024.
11. Spelle L Herbreteau D, Caroff J, et al. CLinical Assessment of WEBTM Device in Ruptured AneurYSms (CLARYS): 12 month angiographic results of a multicenter study. J NeuroIntervent Surg. 2022.
12. Pierot L, Biondi A. Endovascular techniques for the management of wide-neck intracranial bifurcation aneurysms: A critical review of the literature. J Neuroradiol 2016.
13. Rodriguez-Calienes A, Vivanco-Suarez J, Lu Y et al. Woven EndoBridge versus stent-assisted coil embolization for the treatment of ruptured wide-necked aneurysms: A multicentric experience. Interv Neuroradiol 2024.
14. Kortman H, van Rooij S.B.T., Mutlu U. et al. WEB Treatment of Ruptured Intracranial Aneurysms: Long-Term Follow up of a Single-Center Cohort of 100 Patients. American Journal of Neuroradiology 2023.
Indications For Use / Intended Purpose:
TheWEB Aneurysm Embolization System is intended for the endovascular embolization of ruptured and unruptured intracranial aneurysms and other neurovascular abnormalities such as arteriovenous fistulae (AVF). The WEB Aneurysm Embolization System is also intended for vascular occlusion of blood vessels within the neurovascular system to permanently obstruct blood flow to an aneurysm or other vascular malformation. The device should only be used by physicians who have undergone training in all aspects of the WEB Aneurysm Embolization System procedures as prescribed by MicroVention, Inc.
The VIA Microcatheter is intended for the introduction of interventional devices (such as the WEB device/stents/flow diverters) and infusion of diagnostic agents (such as contrast media) into the neuro, peripheral, and coronary vasculature.
Please refer to the IFU for the full list of risks, contraindications, warnings, and precautions.
For Healthcare Professionals Use Only.
![]() WEB™ and VIA™ are trademarks of MicroVention, Inc., registered in the United States and other jurisdictions. All brand names are trademarks or registered trademarks owned by TERUMO CORPORATION, its affiliates, or unrelated third parties.
WEB™ and VIA™ are trademarks of MicroVention, Inc., registered in the United States and other jurisdictions. All brand names are trademarks or registered trademarks owned by TERUMO CORPORATION, its affiliates, or unrelated third parties.
©2025 MicroVention, Inc. M2114(i) EMEA 07/2025
Terumo Neuro Worldwide
Innovation Center
(MicroVention, Inc.)
Legal manufacturer
35 Enterprise
Aliso Viejo, CA 92656 USA
PH +1.714.247.8000
PH +1 800 990 8368
Website : www.terumoneuro.com
Terumo Neuro Europe
(MicroVention Europe S.A.R.L.)
EU Authorized Representative
30 bis rue du Vieil Abreuvoir
78100 Saint-Germain-en-Laye
France
Capital: 40.000€
RCS Versailles 440 775 674
PH +33 (0) 1 39 21 77 46
F +33 (0) 1 39 21 16 01
Terumo Neuro Germany
(MicroVention Deutschland GmbH)
Moskauer Str. 27
D-40227 Düsseldorf
Germany
PH +49 211 210 798-0
F +49 211 210 798-29
Terumo Neuro UK
(MicroVention UK Limited)
Cobalt 13A, Cobalt Park
9 Silverfox Way
Newcastle upon Tyne, NE27 0QJ
United Kingdom
PH +44 (0) 191 258 6777
F +44 (0) 191 258 5999
Terumo Neuro Italy
(MicroVention Italia S.r.l.)
Via Tommaso Gulli, 39
20147 Milano
Italy
PH +39 (0) 02 9475 2414
PH 800 961 631
Terumo Neuro Switzerland
(MicroVention Switzerland GmbH)
Bodenäckerstrasse 3,
CH-8957 Spreitenbach
Switzerland
PH +49 211 / 210 798-0
F +49 211 / 210 798-29
Privacy Policy
Contacts
Terms of Use
Cookies Settings
Hosting by Hubspot

